Posology
VORAXAZE® PRESENTATION1
Voraxaze® 1 000 units powder for solution for injection
White to off-white powder for solution for injection
After reconstitution with 1 mL of sterile 0.9% sodium chloride solution, each vial contains a nominal 1000 units of Voraxaze®†
† Produced in Escherichia coli cells by recombinant DNA technology.
POSOLOGY1
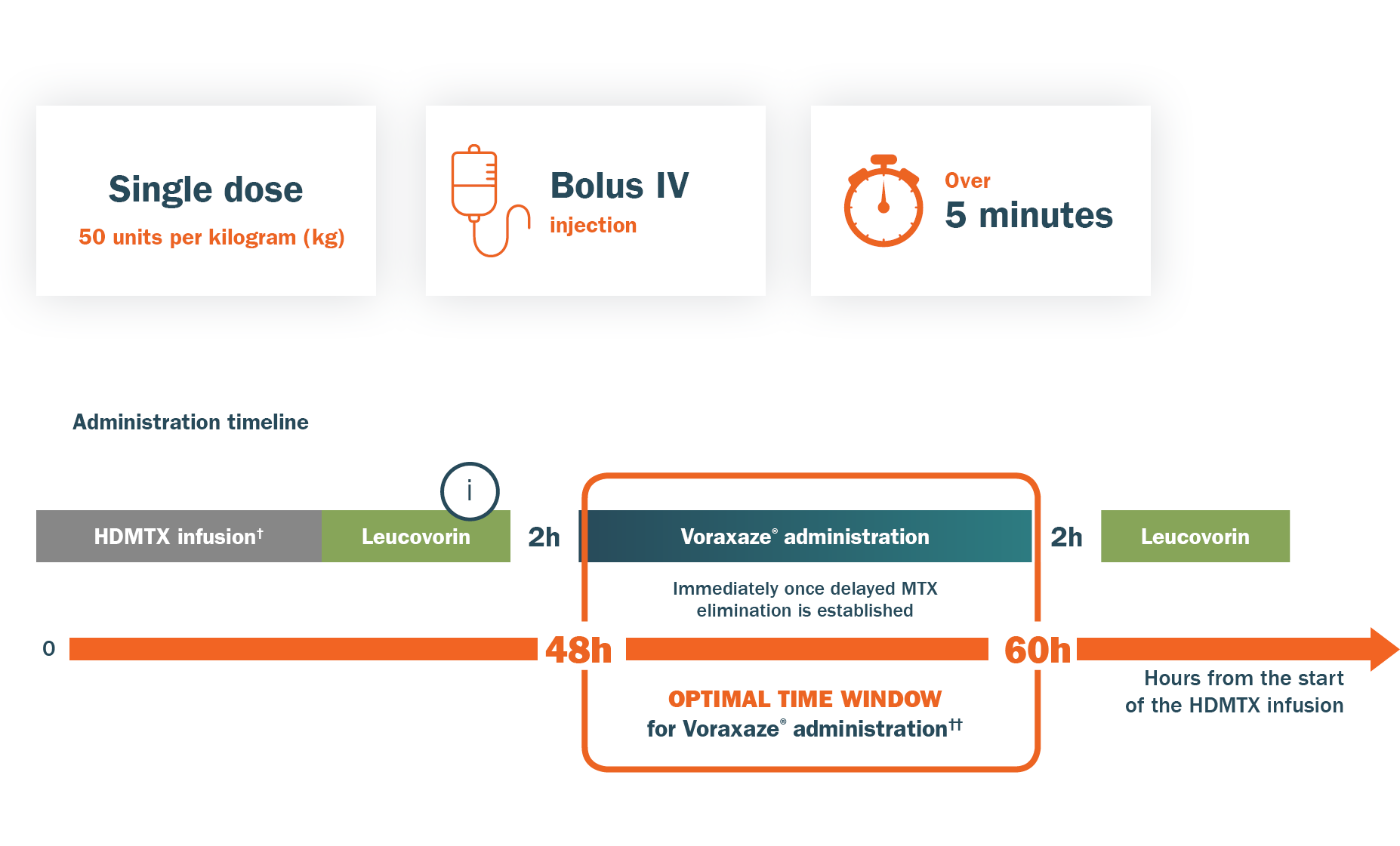
Abbreviations: HDMTX, high dose methotrexate; IV, intravenous; MTX, methotrexate.
†Infusion durations are specific to each protocol and indication (from 2 to 36 hours).2,3
††Clinical data shows Voraxaze® continues to be effective beyond this time window.
ihttps://www.medicines.org.uk/emc/product/9649/smpc/print
References
- Summary of Product Characteristics (SmPC), Voraxaze®.
- Ramsey LB, et al. Consensus guideline for use of glucarpidase in patients with high-dose methotrexate induced acute kidney injury and delayed methotrexate clearance. Oncologist. 2018;23(1):52-61.
- Howard SC, et al. Preventing and managing toxicities of high-dose methotrexate. Oncologist. 2016;21(12):1471-82.
UK-VRX-2600034 Date of Last Revision: February 2026

 United States
United States
 Germany
Germany