Paediatric population
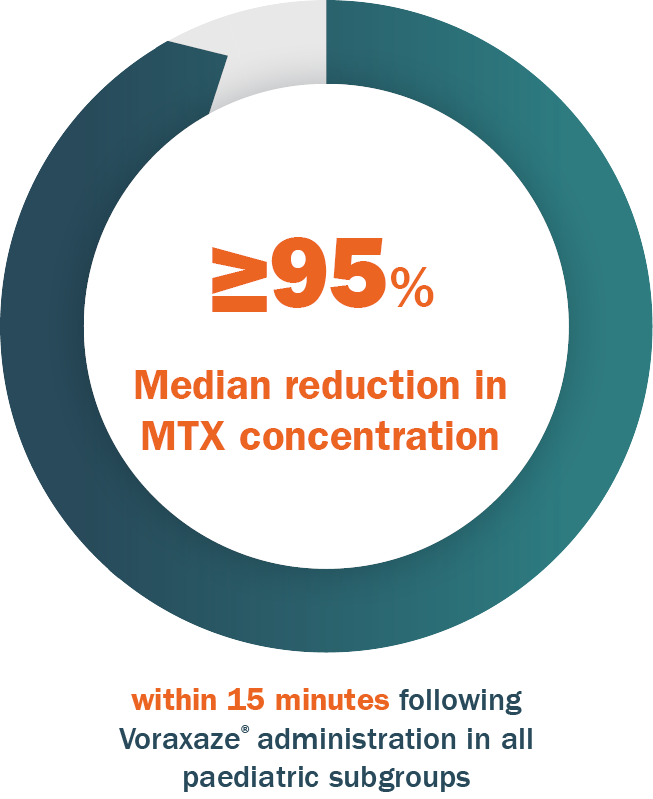
The pooled clinical safety database for Voraxaze® includes data for 232 patients up to 17 years of age1

This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Abbreviations: CIR, clinically important reduction;HPLC, high-performance liquid chromatography; MTX, methotrexate.
References
- Summary of Product Characteristics (SmPC)®.
UK-VRX-2600033 Date of Last Revision: February 2026

 United States
United States
 Germany
Germany