Clinical Studies
FOUR CLINICAL STUDIES to determine the efficacy of VORAXAZE®1,2
Efficacy of Voraxaze® has been evaluated in 4 multi-centre, compassionate use, single-arm, open label studies in patients with delayed MTX elimination. There was no head to head study conducted between these trials - each study was separate and conducted independent of each other.
Patients' characteristics & data available:
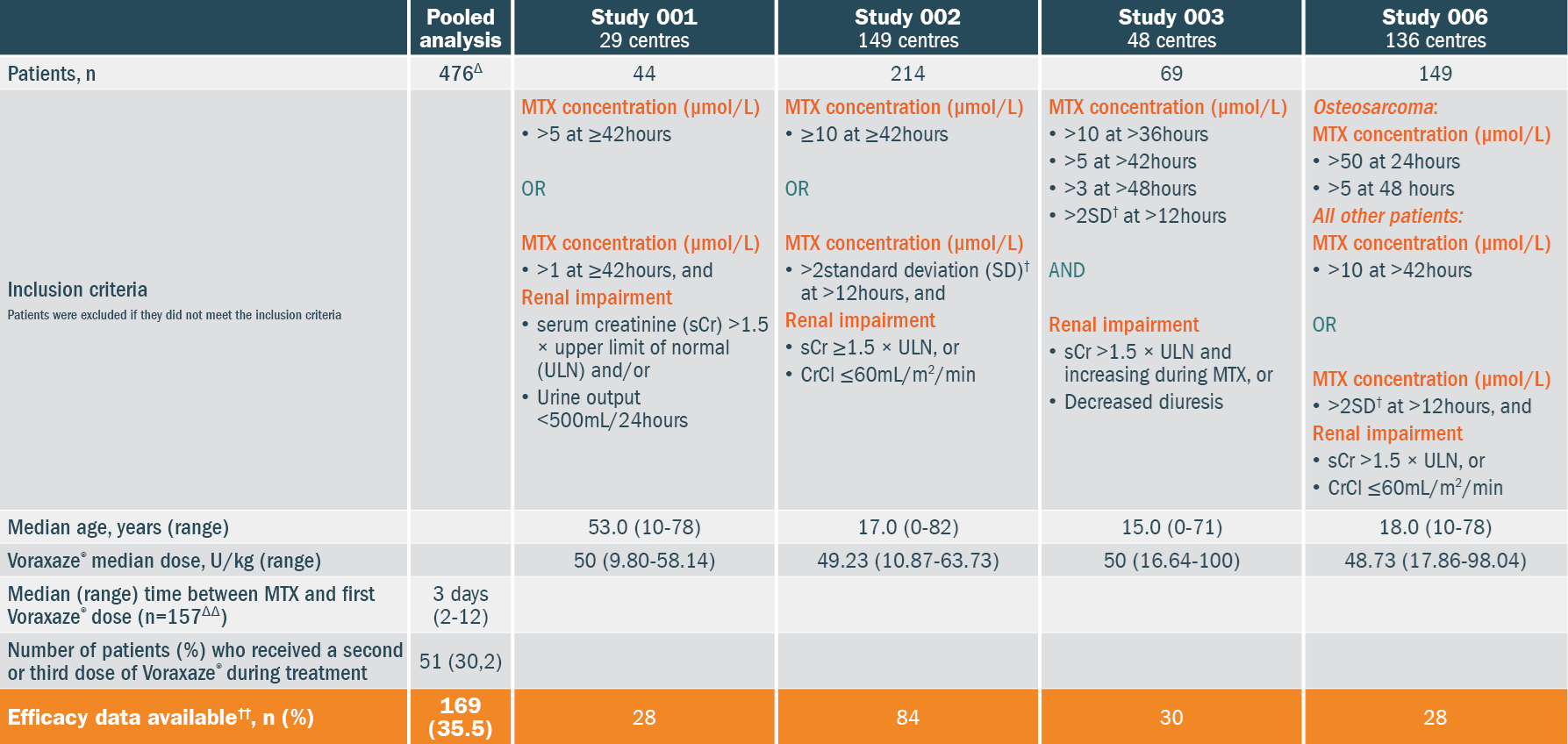
Abbreviations: CrCl, creatinine clearance; MTX, methotrexate; sCr, serum creatinine; SD, standard deviation; ULN, upper limit of normal.
†Plasma MTX concentration >2 SD of the mean MTX excretion curve specific for the dose of MTX administered.
††Patients with at least 1 post-Voraxaze® MTX concentration measured by high-performnance liguid chromatography (HPLC)
ΔPatients with safety data available for analysis in the 4 studies. ΔΔPatients with central MTX date.
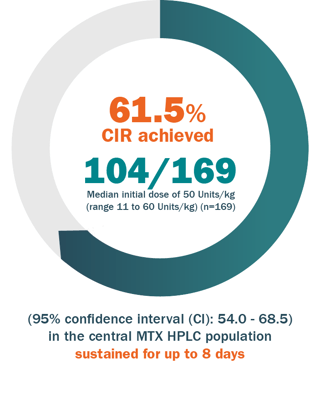
VORAXAZE® allows a CLINICALLY IMPORTANT REDUCTION IN PLASMA MTX CONCENTRATION sustained for up to 8 DAYS1
Efficacy of Voraxaze® has been evaluated in 4 multi-centre, compassionate use, single-arm, open label studies in patients with delayed MTX elimination.
Primary endpoint
Clinically important reduction (CIR) in MTX concentration based on central MTX HPLC data.
- A patient was considered to have achieved a CIR if all central MTX HPLC plasma concentrations after the first dose of Voraxaze® were ≤1 μmol/L
Abbreviations: CIR, clinically important reductions; HPLC, high-performance liquid chromatography; MTX, methotrexate.
VORAXAZE® lowers plasma MTX concentration BY >98% IN 15 MINUTES1,2

Efficacy of Voraxaze® has been evaluated in 4 multi-centre, compassionate use, single-arm, open label studies in patients with delayed MTX elimination.
What is the effect of earlier intervention with Voraxaze® in grade 4 toxicities?3
Prospective study to assess the role of Voraxaze®, leucovorin, and thymidine in the management and outcome of patients with HDMTX-induced nephrotoxicity.
Primary endpoint
Development of grade ≥4 toxicity after Voraxaze® administration.

References
- Summary of Product Characteristics (SmPC), Voraxaze®.
- Widemann BC, et al. Efficacy of glucarpidase (carboxypeptidase G2) in patients with acute kidney injury after high-dose methotrexate therapy. Pharmacotherapy. 2014;34(5):427-39.
- Widemann BC, et al. Glucarpidase, leucovorin, and thymidine for high-dose methotrexate-induced renal dysfunction: clinical and pharmacologic factors affecting outcome. J Clin Oncol. 2010;28(25):3979-86.
UK-VRX-2600033 Date of Last Revision: February 2026

 United States
United States
 Germany
Germany